Ivana Kekez1, Sara Matić2, Mihaela Matovina2, Sanja Tomić2, Dubravka Matković-Čalogović1
1Department of Chemistry, Faculty of Science, University of Zagreb, Zagreb, Croatia
2Divison of Organic Chemistry and Biochemistry, Ruđer Bošković Institute, Zagreb, Croatia
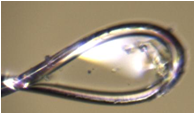
Keap1 is a cellular sensor of oxidative stress that mediates the ubiquitination of the transcription factor Nrf2, targeting it for degradation under basal cellular conditions. However, the Keap1-Nrf2 signalling pathway is impaired in many cancer cells, where expression of Nrf2 target genes leads to resistance to chemotherapeutic drugs. The role of DPP III in oxidative stress was also shown in the study of DPP III KO in mice, where the lack of DPP III was associated with the decrease in Nrf2 activity and increased oxidative stress that caused bone loss due to the increased activity of the osteoclasts [1]. Our group has determined the crystal structure of the Kelch domain of Keap1 in complex with the 11-mer peptide, a fragment of the hDPP III loop containing the ETGE fragment [2]. The next step in studying the influence of mutations related to cancer development was crystallization of the DPP III-Keap1 complex. We performed crystallization trials for the complexes of human DPP III (hDPP III), wild type, as well as its Arg623Trp mutant, and the entire Keap1. The Arg623Trp DPP III mutant was found in cancer samples (https://www.cbioportal.org/) and according to our MST measurements its binding affinity to Kelch is about two orders of magnitude higher than the affinity of the wild type DPP III [2]. This research is designed to increase knowledge of the molecular basis of the hDPP III-Keap1 interaction and its implication on cellular processes and cancer development. After setting 2304 different crystallization conditions using the Oryx8 robot (Douglas Instruments) the best hits were used for further seeding experiments. Of 60 different crystals tested at the XRD2 beamline of the Elettra synchrotron only one diffracted to the resolution of 7 Å with unit-cell parameters of a = b =151.3 Å, c = 221.7 Å, gamma = 120 degree and space group R3 (Fig. 1). Further optimization of crystallization is ongoing and is focused on seeding under new crystallization conditions using the Keap1 construct expressed in insect cells and DPP III treated with inhibitor.
References:
[1] Menale Ciro, Lisa J. Robinson, Eleonora Palagano, Rosita Rigoni, Marco Erreni, Alejandro J. Almarza, Dario Strina, et al. J. Bone Miner. Res. 34 (11) 2019 2133–48. [2] Matić S., Kekez I., Tomin M., Bogár F., Šupljika F., Kazazić S., Hanić H., Jha S., Brkić H., Bourgeois B., Madl T., Gruber K., Macheroux P., Matković-Čalogović D., Matovina M. & Tomić S. J. Biomol. Struct. Dyn. 39:18 2020 6870-6881.
Keywords: crystallization; seeding; Keap1; DPP III; complex
